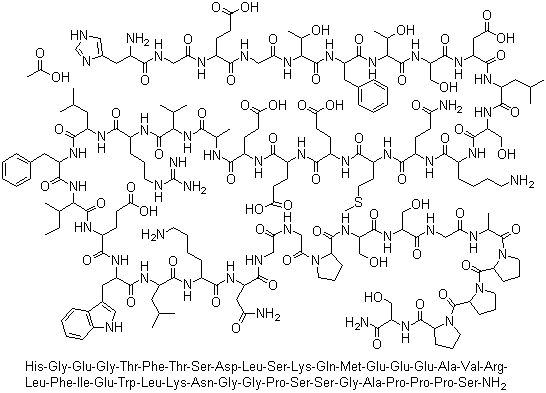
141758-74-9
- Product Name:Exenatide acetate
- Molecular Formula:C184H282N50O60S
- Purity:99%
- Molecular Weight:4186.57
Product Details;
CasNo: 141758-74-9
Molecular Formula: C184H282N50O60S
Quality Manufacturer Supply Exenatide acetate, Factory Sells 141758-74-9 Competitive Price
- Molecular Formula:C184H282N50O60S
- Molecular Weight:4186.57
- PSA:1775.05000
- LogP:-2.16940
Exendin-4(Cas 141758-74-9) Usage
|
New diabetes drugs |
Diabetes is a metabolic disorder characterized by chronic hyperglycemia caused by a variety of causes , high blood sugar is caused by insulin secretion or its effect defect. Diabetes can be divided into type 1 diabetes, type 2 diabetes, other specific types of diabetes, and gestational diabetes, among which type 2 diabetes account for more than 90%. Exendin-4 is a new diabetes drug successfully developed by American Eli Lilly Company it belongs to incretin analogues, it is the first member of the incretin analogues family , it is the synthetic peptide compound, which can simulate physiological behavior of the natural state secretion of GLP-1 in vivo ,it is similar to human pancreatic glucagon-like peptide effect-1 (GLP-1),it can promote glucose-dependent insulin secretion, it has inhibition effect of inappropriate glucose-dependent glucagon secretion, slowing gastric emptying, it improves the sensitivity of peripheral tissues to insulin, and it makes blood sugar adequately controlled. Clinically it is used for the treatment of type Ⅱ diabetes patients whose blood sugar is unbale to be controlled by metformin, sulfonylurea, or combination of metformin and sulfonylurea. From April 28, 2005 to October 29, 2008, the US Food and Drug Administration (abbreviation: FDA) Adverse Event Reporting System had received reports of 78 cases of patients with renal exenatide change. In the meantime, the United States had made a total of more than 6.6 million Exendin-4 prescription, therefore, FDA thought the received 78 cases was in a small proportion of the proportion of all patients reporting use of the drug. Currently FDA has completed an assessment of these reported cases, including 62 cases of acute renal failure cases and 16 cases of renal insufficiency cases. Acute renal failure or renal dysfunction occurs three days to two years after treatment. The age span is 23 years to 83 years, mean age is 60 years. |
|
Uses |
Exenatide is indicated as adjunctive therapy to improve glycemic control in patients with type 2 diabetes who are taking metformin, a sulfonylurea, or both but have not achieved adequate glycemic control. It activates the GLP-1 receptor, increasing cAMP levels in pancreatic acinar cells without affecting the VIP receptor. Exenatide moderates peak serum glucagon levels during hyperglycemia after meals and does not interfere with glucagon release in response to hypoglycemia. Administered as a subcutaneous injection, the dosing regimen is typically 5 or 10 mg twice daily, within an hour before morning and evening meals. Common adverse effects include nausea, vomiting, diarrhea, jitteriness, dizziness, headache, and dyspepsia. |
|
Description |
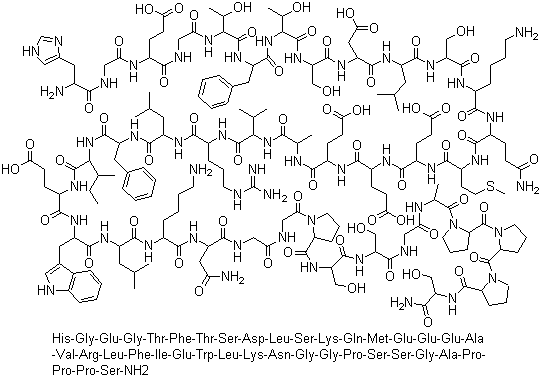
Exenatide acetate is a medication classified as an incretin mimetic, specifically a functional analog of the human incretin Glucagon-Like Peptide-1 (GLP-1). GLP-1 is a natural hormone released in response to food intake, and it acts on receptors to enhance glucose-stimulated insulin secretion. Exenatide is a synthetic version of a 39-amino acid peptide found in the salivary secretions of the Gila monster lizard. Its long-acting agonist properties make it a novel option for diabetes management. |
|
Originator |
Amylin (US) |
|
Definition |
ChEBI: A bioactive polypeptide of 39 amino acid residues isolated from the saliva of the Gila monster (Heloderma suspectum). High-affinity glucagon-like peptide 1 (GLP-1) receptor agonist (Kd = 136 pM); potently indu es cAMP formation without stimulating amylase release in pancreatic acini; potentiates glucose-induced insulin secretion in isolated rat islets; protects against glutamate-induced neurotoxicity. A synthetic version is called exenatide. |
|
Brand name |
Byetta |
|
Biochem/physiol Actions |
Activates GLP-1 (glucagon-like peptide-1) receptors to increase intracellular cAMP in pancreatic acinar cells; has no effect on VIP receptors. |
|
in vivo |
in ob/ob mice, administration of exendin-4 (10 μg/kg or 20 μg/kg) improved insulin sensitivity and significantly reduced serum glucose and hepatic steatosis. exendin-4 appeared to effectively reverse hepatic steatosis in ob/ob mice by improving insulin sensitivity [1]. in athymic mice, 63% of exendin-4-treated mice achieved graft function compared with 21% of untreated mice (p = 0.033) in the short-term study. 88% of treated mice had functioning grafts compared with 22% of controls (p = 0.015) in the long-term study. exendin-4-treated mice gained significantly more weight than the untreated counterparts [4]. |
|
references |
ding x, saxena n k, lin s, et al. exendin‐4, a glucagon‐like protein‐1 (glp‐1) receptor agonist, reverses hepatic steatosis in ob/ob mice[j]. hepatology, 2006, 43(1): 173-181.gke r, fehmann h c, linn t, et al. exendin-4 is a high potency agonist and truncated exendin-(9-39)-amide an antagonist at the glucagon-like peptide 1-(7-36)-amide receptor of insulin-secreting beta-cells[j]. journal of biological chemistry, 1993, 268(26): 19650-19655.perry t a, haughey n j, mattson m p, et al. protection and reversal of excitotoxic neuronal damage by glucagon-like peptide-1 and exendin-4[j]. journal of pharmacology and experimental therapeutics, 2002, 302(3): 881-888.sharma a, srenby a, wernerson a, et al. exendin-4 treatment improves metabolic control after rat islet transplantation to athymic mice with streptozotocin-induced diabetes[j]. diabetologia, 2006, 49(6): 1247-1253. |
InChI:InChI=1S/C184H282N50O60S/c1-16-94(10)147(178(289)213-114(52-58-144(257)258)163(274)218-121(73-101-77-195-105-39-24-23-38-103(101)105)168(279)215-116(68-90(2)3)165(276)205-107(41-26-28-61-186)158(269)219-122(75-134(189)243)154(265)198-79-135(244)196-83-139(248)231-63-30-43-129(231)175(286)225-127(87-238)174(285)223-125(85-236)155(266)200-80-136(245)202-96(12)181(292)233-65-32-45-131(233)183(294)234-66-33-46-132(234)182(293)232-64-31-44-130(232)176(287)222-124(84-235)150(190)261)229-170(281)119(71-99-34-19-17-20-35-99)217-166(277)117(69-91(4)5)214-159(270)108(42-29-62-194-184(191)192)212-177(288)146(93(8)9)228-151(262)95(11)203-156(267)111(49-55-141(251)252)208-161(272)112(50-56-142(253)254)209-162(273)113(51-57-143(255)256)210-164(275)115(59-67-295-15)211-160(271)110(47-53-133(188)242)207-157(268)106(40-25-27-60-185)206-172(283)126(86-237)224-167(278)118(70-92(6)7)216-169(280)123(76-145(259)260)220-173(284)128(88-239)226-180(291)149(98(14)241)230-171(282)120(72-100-36-21-18-22-37-100)221-179(290)148(97(13)240)
Relevant Products
-
Exenatide acetate Powder
CAS:141732-76-5
-
Carperitide
CAS:89213-87-6
-
Bremelanotide acetate
CAS:1607799-13-2